Graphene (GFET) Chemical Sensors
From toxic leaks in industrial facilities to administering drugs in medical clinics, there is always a real need to quickly and effectively detect chemicals which are normally invisible to the naked eye. Huge strides in chemical sensing technology over recent years have enabled more efficient detection of lower concentrations of substances in many different environments.
Graphene, with its unique qualities, has proved to be the ‘go-to’ material for researchers in this area; vastly improving the performance of chemical sensors in ways other materials cannot.
What Are Chemical Sensors?
Simply put, a chemical sensor is a device which can detect an individual target chemical and turn this interaction into a measurable output. In an ideal world, these devices would be cheap to make, portable, and easy to use, responding instantly with perfect precision when interacting with a particular chemical compound at any concentration. While technology is continuously advancing with regards to the effectiveness of chemical sensors however, there is still work to be done before we can reach this ideal type of device.
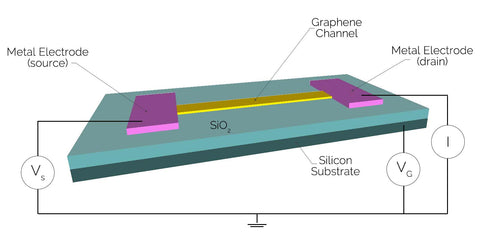
In reality, chemical sensors are more complicated, having to be optimised and adapted to a specific type of application. While sensors come in a variety of types, they can be split into two categories:
- Direct-reading, selective sensors such as electrochemical sensors and optical fibres
- Sensors which contain an initial column chromatographic or capillary electrophoretic sample separation step followed by a detection which is sensitive but not necessarily selective
As a wider variety of sensors are developed and further research is conducted to make these devices even more effective, the market is also expanding to keep up. In 2017, the global chemical sensor market was valued at US$18.56 billion. This is expected to reach US$28.16 billion by 2023, at a compound annual growth rate (CAGR) of 7.20%.
Graphene and Chemical Sensors
In the world of chemical sensors, graphene has attracted a lot of attention, not least because its two-dimensional structure means the entire material volume acts as a sensor surface. Furthermore, graphene provides excellent mechanical strength, thermal and electrical conductivity, compactness, and a potentially low cost, which is necessary for competing in the ever-growing chemical sensor market.

The most simple and common configuration for graphene-based chemical sensors is the graphene field-effect transistor (GFET), a sheet of graphene with a sensing area between two metal contacts. In this device, the conductivity can be tuned using the electric field effect with a back gate, yielding a very high sensitivity. Carrier mobility should be high to ensure minimal losses to heating. Graphene is a prime candidate for creating superior GFETs as chemical sensors because the material itself is biocompatible, can be used unprotected in ambient circumstances, has ultra-high mobility, and low resistivity. This means more efficient chemical sensors which will lead to superior computers and electronic measuring devices.

Whether as GFETs or utilizing other designs, graphene-based gas and vapor sensors have held the spotlight in recent years due to their variety of structures, unique sensing performance, room-temperature working conditions, and tremendous application prospects. Apart from water vapor, graphene has been used to sense gases such as NH3, NO2, H2, CO, SO2, H2S, as well as vapor of volatile organic compounds, resulting in a dramatic rise in scientific publication numbers on this topic. Graphene has also been used to detect traces of opioids in concentrations as low as 10 picograms per milliliter of liquid.
Manufacturing Graphene Chemical Sensors
Generally, graphene is produced by chemical vapor deposition (CVD). This process is a way to deposit gaseous reactants onto a catalyst substrate. By first combining gas molecules in a gas chamber, a material film can be formed when the combined gases come into contact with the substrate itself. The waste gases are then pumped from the chamber. The temperature is the key condition here, determining the type of reaction that occurs and leading to a successfully produced film. There are a few difficulties with CVD however, including separation of the graphene film from the substrate and creating a uniform layer of graphene on the substrate.
Heightened Sensitivity for Better Detection
The simplicity of GFETs and the ability to tune the conductivity have seen great advances in optimizing the sensitivity of these devices as sensors. For instance, researchers from the University of Manchester showed these sensors could detect a single gas molecule as it attached to or detached from the graphene surface. This sensitivity is due to the fact that the device’s electrical resistance changes when something attaches to the graphene. Certain gases have shown up to 15% change in resistance, with others like methanol showing an easily detectable change of around 5%.
CVD graphene sensors made in Northwestern University, Illinois have shown detection limits for ammonia (NH3) on the ppb level, superior to commercially available devices. Detection of NH3 is essential because, although widely used in the creation of pharmaceuticals and many commercial products, it is caustic and hazardous, harmful to humans, with the potential to cause environmental pollution. GFETs have also been used to detect nitrogen dioxide (NO2), another industrially relevant gas harmful to humans and the environment. A team at Shanghai Jiao Tong University developed NO2 sensors based on graphene with detection limits below ppm, high sensitivity, as well as excellent selectivity and response speed.
All other graphene-based chemical sensors, such as those for hydrogen gas, carbon dioxide, carbon monoxide, methane, and sulfur dioxide, show detection limits better or on par with commercially available sensors.

Figure: Sensitivity of graphene sensors to various gases (Sens. Actuators B 163(1), 107–114 (2012)).
Bigger, better, bolder
Because of its physical properties, high-quality CVD graphene is a prime candidate for development in many areas. One of these is the fabrication of large-area, commercially viable GFET sensors. In 2015, scientists from the University of Glasgow managed to fabricate large area graphene sheets of approximately 400 cm2 using standard copper foils in CVD, utilizing hot lamination to transfer the graphene to a PVC substrate.
Research is also being done to expand the functionality of graphene-based chemical sensors ranging from chemical-sensitive modulators for use in internet-of-things applications to creating wearable electronics for use in the hospital, on the battlefield and more. A team from the Barcelona Institute of Science and Technology has used graphene to create high resolution broadband image sensors while scientists at Hanyang University in Korea have found a way to use graphene sensors in virtual reality devices to feel and distinguish between different surface textures. With all of this happening over the past few years, we can only imagine where the world of graphene chemical sensors will take us next.
Graphenea Offer A Range of Graphene-Based Field Effect Transistors in different configurations for research purposes, and now also offer custom graphene device printing (GFAB). See the full range here, or contact us today to discuss any specific configurations you need.
